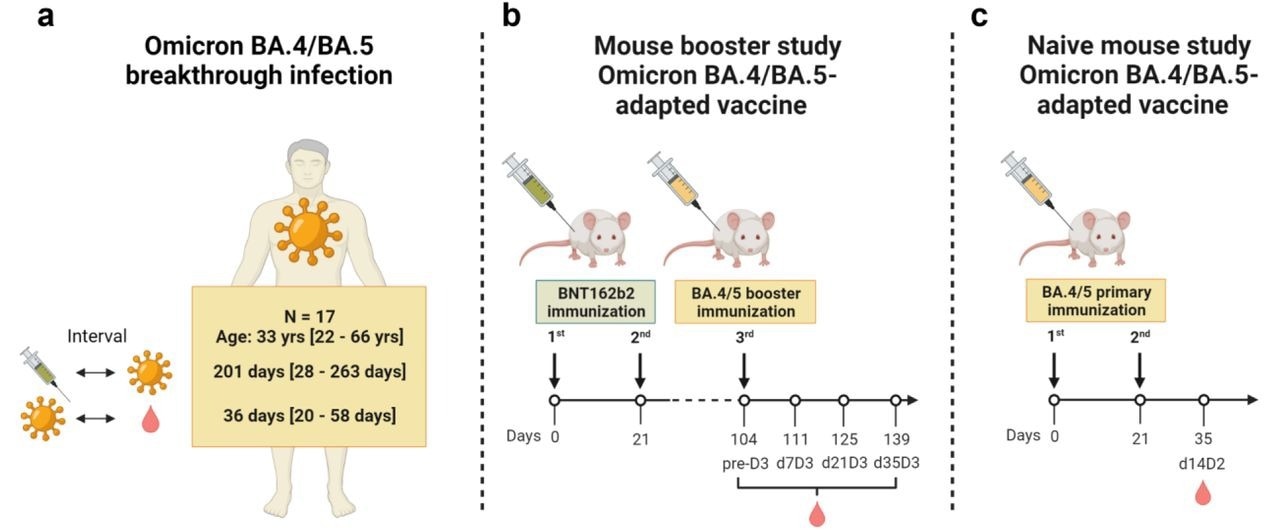
Omicron BA.4/BA.5-adapted bivalent vaccine generates higher antibody response - Hospital Pharmacy EuropeHospital Pharmacy Europe

News - CHMP Recommends Additional Authorisation Modification of Comirnaty (BioNTech/Pfizer) As a Bivalent Vaccine Adapted to Omicron BA.4/BA.5 for Booster Vaccinations - Paul-Ehrlich-Institut
Covid: Aifa approva il vaccino bivalente Comirnaty sviluppato contro Omicron 4-5 | Sanità24 - Il Sole 24 Ore
News - CHMP Recommends Authorisation of Comirnaty Variant (BioNTech/Pfizer) Adapted to Omicron BA.4/BA.5 As Booster Vaccination for Children Aged 5 to 11 Years - Paul-Ehrlich-Institut

Pfizer Canada - Another step forward in the fight against COVID-19: Health Canada has authorized COMIRNATY Original & Omicron BA.4/BA.5 (Pfizer-BioNTech bivalent, Omicron BA.4/BA.5-adapted COVID-19 vaccine) as a booster dose for Canadians
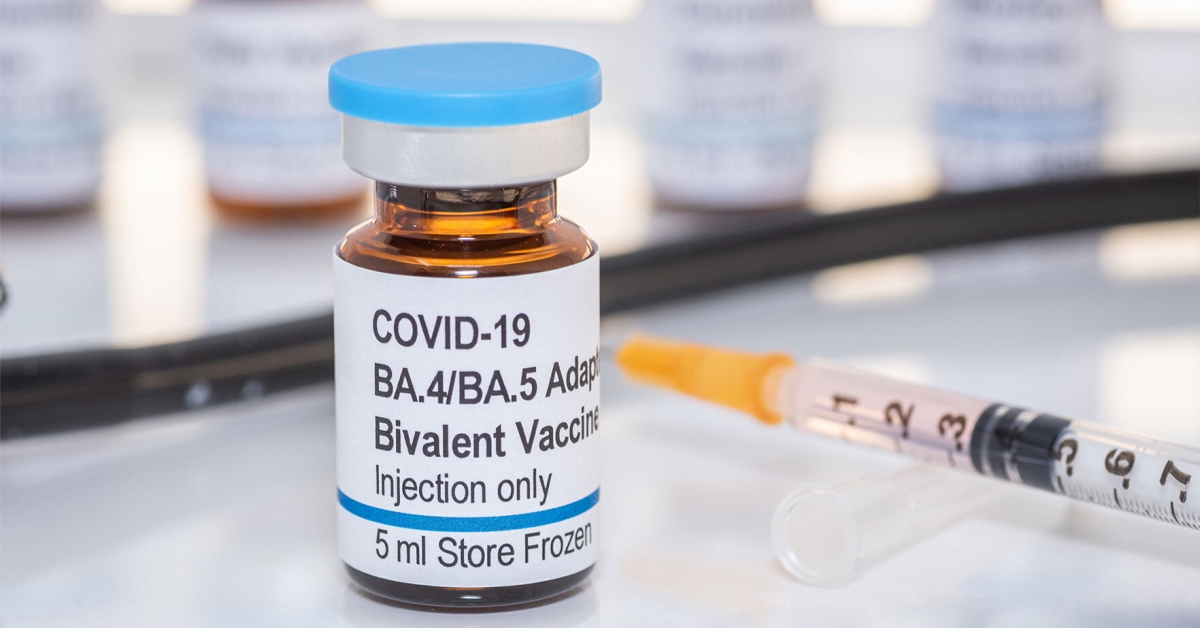
Common Questions About Bivalent COVID-19 Boosters | Johns Hopkins | Bloomberg School of Public Health